Book Appointment Now
Your Peptide Research Partner in Europe
Pharmalyx-Europe is dedicated to peptide research and education, with a strong focus on GLP-1–related peptides. Our content is developed for scientific, educational, and research purposes in line with EU compliance standards.
Peptide Science & Research Context
An overview of peptide biology, GLP-1 research focus, and investigational developments such as Retatrutide.

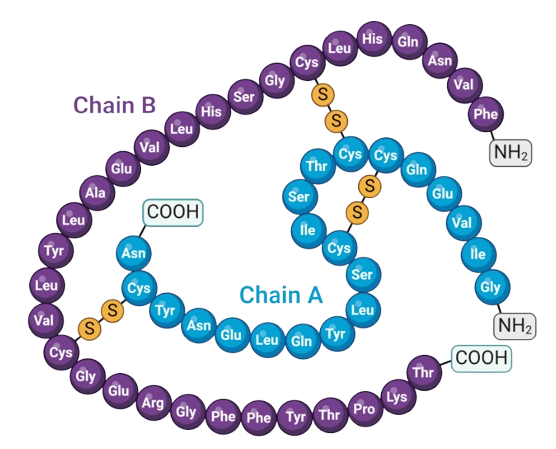
What Are Peptides?
Peptides are short chains of amino acids that play a critical role in biological signaling and metabolic processes. They act as messengers in the body, influencing functions such as appetite regulation, glucose metabolism, and energy balance.

Our Research Focus
Our educational content centers on GLP-1–related peptides, presented within a scientific and research-driven framework. We aim to provide accurate, up-to-date information for professionals and researchers interested in peptide science.
Research Background
Retatrutide is an investigational compound currently being studied for its potential role in obesity and type 2 diabetes research. It is designed to support weight reduction and improve metabolic parameters. At present, Retatrutide remains in clinical development and is not approved for general medical use.

Triple Hormone Agonist Mechanism
Retatrutide acts as a triple hormone receptor agonist, targeting:
- GLP-1 receptor – appetite regulation and glucose control.
- GIP receptor – insulin response modulation.
- Glucagon receptor – energy expenditure and metabolism.
By influencing all three pathways, Retatrutide is being studied for its potential to reduce hunger, enhance satiety, increase metabolic activity, and support weight management.
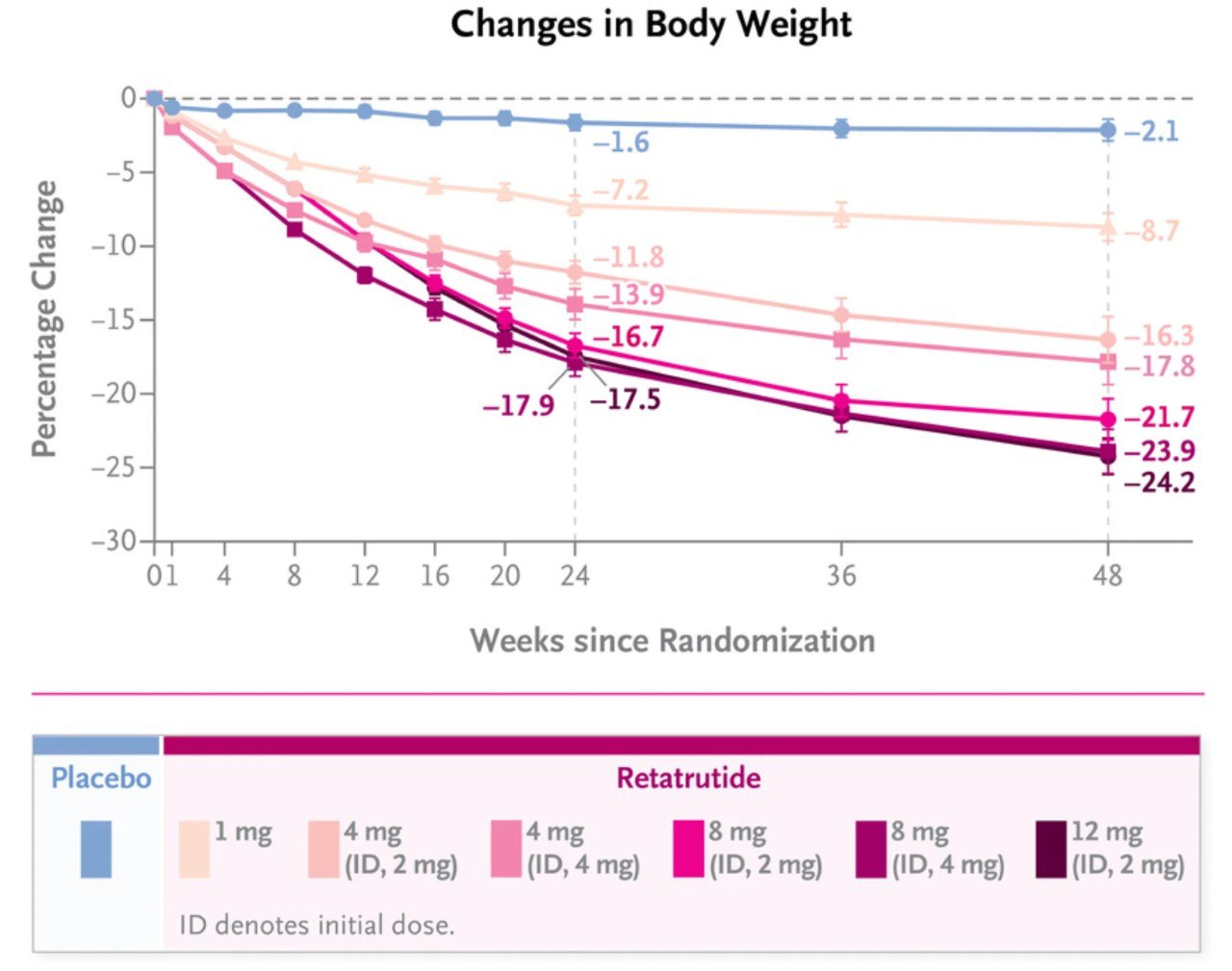
Current Research Insights
Clinical studies have reported significant weight-loss outcomes, in some cases exceeding those observed with compounds such as Semaglutide and Tirzepatide. These findings position Retatrutide as a potential next-generation peptide under investigation.
Note: Further studies are ongoing to better understand long-term outcomes, safety, and efficacy.

Safety Profile (Research Context)
Reported side effects in clinical settings have generally been mild and most commonly observed during the initial phase of treatment. Ongoing trials continue to evaluate tolerability and safety parameters.

Research Materials
- Retatrutide – 10 mg
- High-purity research material
- For laboratory and research use only

Important Notice
This website and its content are intended for research and educational purposes only. Information provided does not constitute medical advice, diagnosis, or treatment recommendations.
